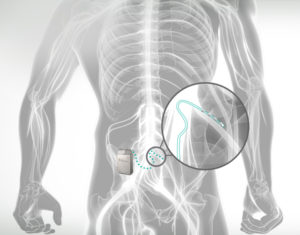
A person’s source of pain and how to target it has always been the focal point of pain intervention. Fortunately in recent years, a popular and effective way for those who have not found relief for their complex regional pain syndrome (CRPS) with traditional spinal cord stimulation (SCS) or other treatments has emerged. Recently, an advanced treatment has emerged that is the stimulation of the neural structure called the dorsal root ganglion (DRG), The DRG consists of clusters of nerve cells stemming from your spine. These nerve structures serve as “highways of pain signals” and are critical in the communication of pain from your body to your brain. DRG neurostimulation has the capability to positively modify the pain signals that transmit through these clusters. This is all possible after the implantation of a device that extends an electrical lead to the location of the disrupted dorsal root ganglion. The device produces electrical signals which modify the pain pathways to reduce pain and the signs and symptoms associated with CRPS (formally known as reflex sympathetic dystrophy – RSD). Signs and symptoms of Complex Regional Pain Syndrome may include color, sweating and temperature differences. Dorsal root ganglion stimulation offers advantages over traditional SCS for specific pain conditions due to the specific placement of these leads directly over the DRG allowing for specific targeting of the painful area, paresthesia (i.e. tingling or buzzing sensation associated at the pain site or non-targeted areas), reduced stimulation and less influence of position on stimulation. The ACURRATE study, demonstrated that 86% of patients receiving DRG stimulation at 12 months had greater than 50% improvement in pain scores and freedom from a stimulation related neurological deficit. The implanted device also uses less energy which minimizes battery maintenance including recharging. The innovation DRG stimulation has resulted in significant advancement in the treatment of neuropathic pain including CRPS. As with other neuromodulation devices, patients are able to undergo a trial phase that helps determine if the therapy is correct prior to undergoing implantation stage.
Acknowledgment: Axium and St. Jude Medical are trademarks of St. Jude Medical, Inc. or its related companies. Reproduced with permission of St. Jude Medical, ©2016. All rights reserved.
Contact form
Or just call (412) 221-7640 and we’ll do everything we can to help.
Across all review platforms
Dr. David Provenzano, a leading expert throughout the nation for treating patients who suffer daily from pain, uses advanced diagnostic techniques to assess the source of the pain and develop a comprehensive and safe treatment approach that can significantly reduce your pain.

